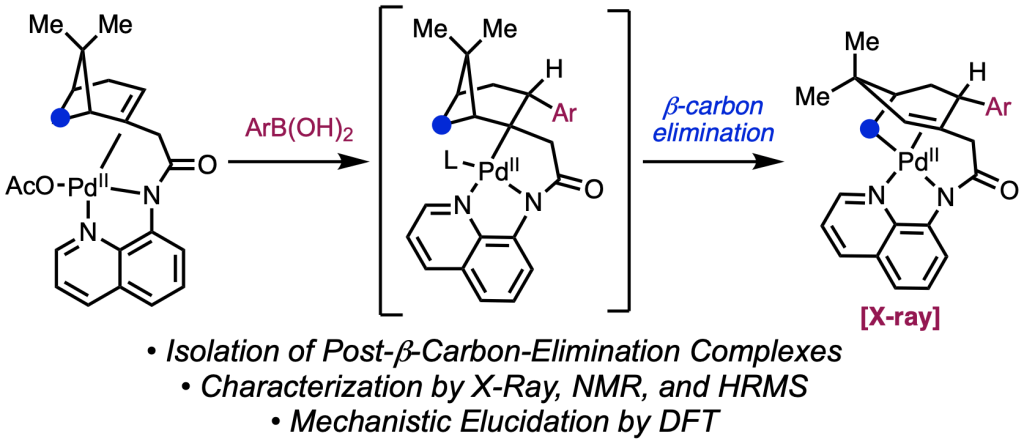
Today we report a new strategy for alkene functionalization that can deliver two valuable products in a single step by partnering a mutually enabling oxidative difunctionalization and reductive difunctionalization, a process we term redox-paired alkene functionalization. In the reported reaction system, a single palladium(II) catalyst switches between PdIIX and PdIIAr forms during the catalytic cycle, each of which is active in the respective half-reactions. Specifically, the catalytic cycle achieves oxidative [3+2] annulation with 2-naphthols and reductive 1,2-diarylation with aryl iodides, expanding the scope and utility of both of these transformations. Congrats to all of the co-workers in this collaborative project: Hui-Qi, Tian (May), Shenghua, and Zhen; as well as Malkanthi, who performed computational studies under the joint guidance of Profs. Ken Houk (UCLA) and Peng Liu (Pittsburgh).
For a link to the pre-print in ChemRxiv, click here: https://chemrxiv.org/engage/chemrxiv/article-details/63ad635fff4651a6983afddf