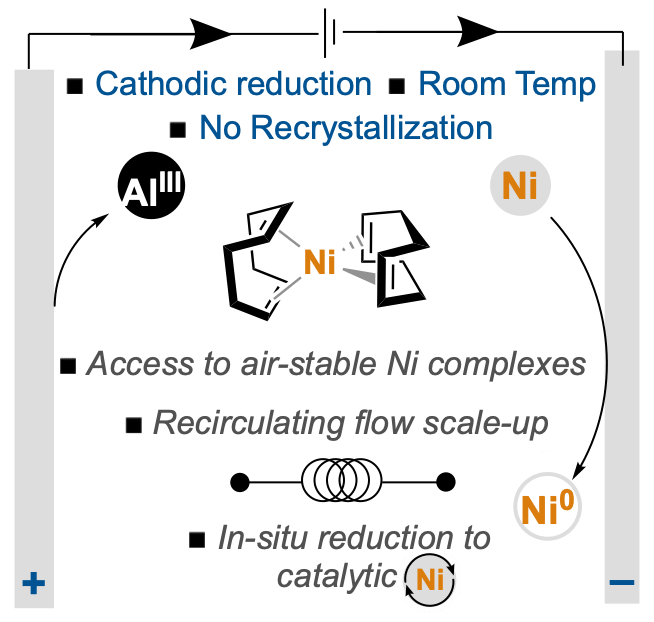
The final version of our manuscript describing an electrochemical method to covert NiII salts into various synthetically useful Ni0 complexes appears today in Angewandte Chemie International Edition. Through systematic optimization of reaction parameters, we developed a method that avoids traditional stoichiometric organometallic reductants, such as DIBAL–H, that are commonly employed in this process. The method is robust, scalable via a recirculating flow setup, and applicable to numerous important products (e.g., Ni(COD)2, Ni(COD)(DQ), and Ni(PP3)4). Congrats to the entire collaborative team, including Camille and Yilin from our lab, Tamara and Gabriele from the Baran lab at Scripps, Greg and Steve from Bristol Myers Squibb, Xiangyu from the Lin lab at Cornell, and Julien from Syngenta/University of Lyon.
For a link to the paper in Angew. Chem. Int. Ed., click here: https://onlinelibrary.wiley.com/doi/abs/10.1002/anie.202311557
As a reminder, a ChemRxiv pre-print disclosing this work first appeared back in April: https://chemrxiv.org/engage/chemrxiv/article-details/642b6699a029a26b4cde3e3e