Check out commercially available catalysts, ligands, and reagents from the Engle lab: MilliporeSigma Professor Product Portal
Overview: Organic small molecules make up the vast majority of medicines, biological probes, agrochemicals, and materials building blocks. Thus, though they are ubiquitous in modern society, many classes of small molecules remain problematic to prepare using conventional methods. Contemporary synthetic routes routinely require multiple steps, generate large quantities of waste, or require substantial human energy to troubleshoot. Research in the Engle group focuses on addressing these challenges by harnessing the power of catalysis. Our group’s goal is to advance the efficiency, effectiveness, and sustainability of chemical synthesis. The projects that we tackle are highly interdisciplinary, and as a consequence, students complement their core training in synthetic organic, physical organic, and organometallic chemistry with expertise in related disciplines in and outside of chemistry.
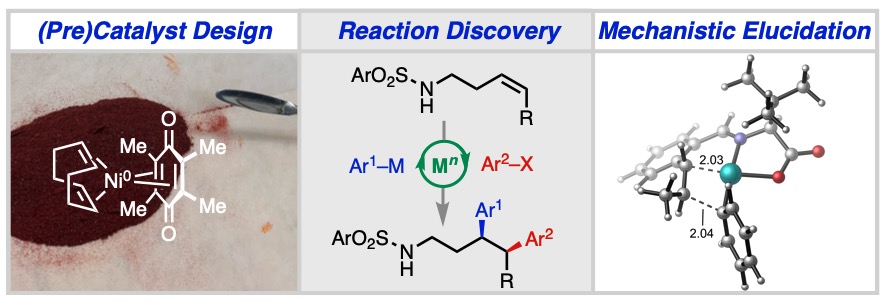
1. (Pre)Catalyst Design: A principal driver of innovation in homogeneous catalysis is the design of ancillary ligands that can imbue the metal center with desired combination of reactivity, selectivity, and stability. As chemists, ligands are our “hands at the transition state” allowing us to manipulate intermediate and transition state stabilities with molecular-level control. In the Engle lab, we approach ligand and (pre)catalyst design by combining organometallic synthesis, structural characterization, and DFT calculations.
For a representative research publication, see:
(a) Tran, V. T.; Li, Z.-Q.; Apolinar, O.; Derosa, J.; Wisniewski, S. R.; Joannou, M. V; Eastgate, M. D.; Engle, K. M. “Ni(COD)(DQ): An Air-Stable 18-Electron Ni(0)–Olefin Precatalyst,” Angew. Chem. Int. Ed. 2020, 59, 7409.
2. Reaction Discovery: Owing to their energetically accessible d-orbitals, transition metals possess a unique ability to mediate bond-making and bond-breaking events that are otherwise foreign to main group elements. By leveraging transition metal catalysis, it is thus possible to rapidly generate structurally complex and functionally diverse small-molecule products from chemical feedstocks. In the Engle lab, reaction discovery hinges on hypothesis-driven experimentation, high-throughput screening, and (of course!) chemical intuition.
For representative reviews, see:
(a) Derosa, J.; Apolinar, O.; Kang, T.; Tran, V. T.; Engle, K. M. “Recent Developments in Nickel-Catalyzed Intermolecular Dicarbofunctionalization of Alkenes,” Chem. Sci. 2020, 11, 4287.
(b) Liu, Z.; Gao, Y.; Zeng, T.; Engle, K. M. “Transition-Metal-Catalyzed 1,2-Carboboration of Alkenes: Strategies, Mechanisms, and Stereocontrol,” Isr. J. Chem. 2020, 60, 219.
3. Mechanistic Elucidation: Understanding the details of how a given catalytic cycle functions is vital for: (a) troubleshooting issues when reactivity goes awry; (b) controlling regio-, chemo-, stereo-, and pathway-selectivity; and (c) developing improved ligands, catalysts, and reagents. By drawing on a wide range of techniques from computational chemistry (density functional theory, DFT) to reaction kinetics (reaction progress kinetic analysis, RPKA) and working closely with collaborators with complementary expertise in these and other areas, the Engle lab endeavors to obtain a comprehensive understanding of newly developed reactions.
For representative research publications, see:
(a) Vasquez, A. M.; Gurak, J. A., Jr.; Joe, C. L.; Cherney, E. C.; Engle, K. M. “Catalytic a-Hydroarylation of Acrylates and Acrylamides via an Interrupted Hydrodehalogenation Reaction,” J. Am. Chem. Soc. 2020, 142, 10477.
(b) Romine, A. M.; Yang, K. S.; Karunananda, M. K.; Chen, J. S.; Engle, K. M. “Synthetic and Mechanistic Studies of the a Versatile Heteroaryl Thioether Directing Group for Pd(II) Catalysis,” ACS Catal. 2019, 9, 7626.
