We are delighted to share our new review article, “Catalytic Functionalization of Unactivated π-Bonds Enabled by Bidentate Directing Auxiliaries,” now published in Angewandte Chemie International Edition!
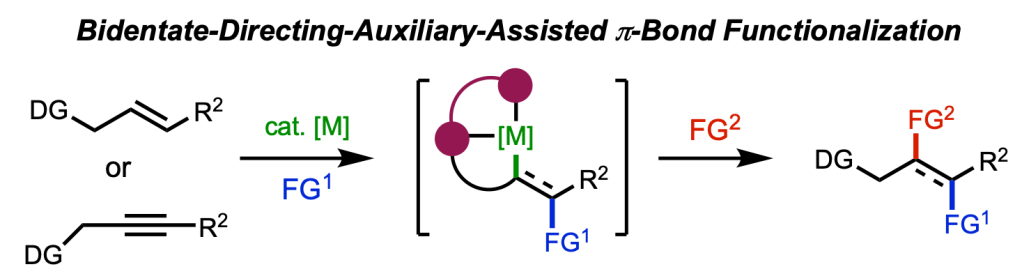
A defining theme of our group’s research over the past decade has been the use of bidentate directing auxiliaries to enable new catalytic functionalization reactions of π-bonds and to facilitate mechanistic elucidation. It has been gratifying to watch this approach gain widespread adoption across the synthetic community.
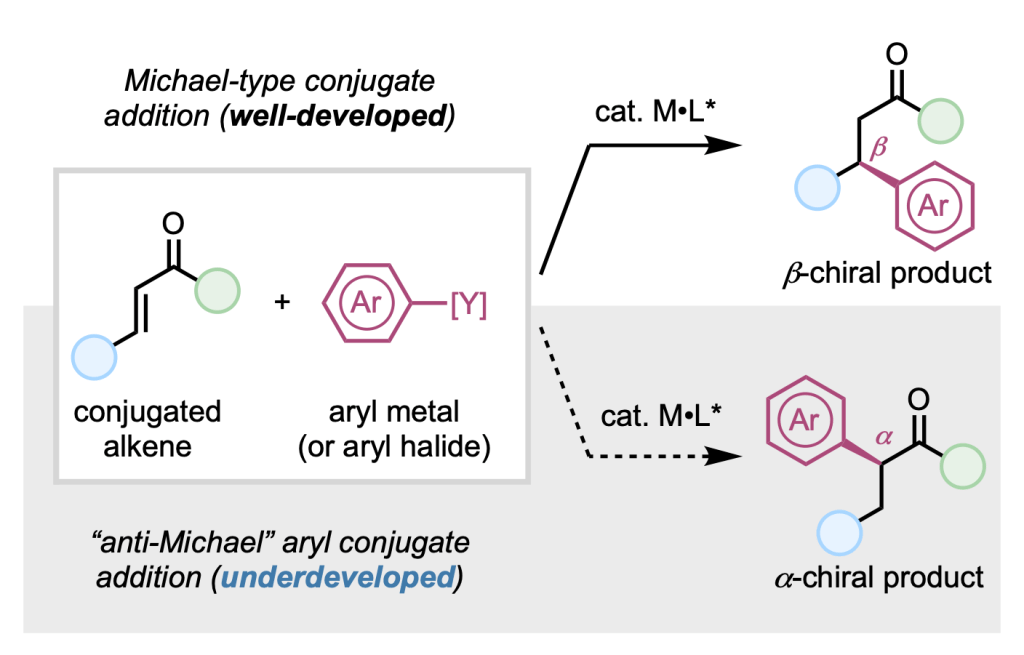
In this review, we join forces with the laboratory of Prof. Taeho Kang at Korea University to take stock of where the field stands — covering key developments in hydrofunctionalization, difunctionalization, and C–H activation across palladium, nickel, copper, and other transition-metal catalysts, and discussing the outstanding challenges and opportunities that will shape the next chapter of this chemistry.
Congratulations to all of the student coauthors: Seunghyeon Cho (Korea University), Al Vicente Riano D. Lisboa (Scripps Research), and Juntao Sun (Scripps Research). Thank you to Prof. Kang and the whole team for such a wonderful collaboration!
📄 Read the review in Angewandte Chemie International Edition: https://onlinelibrary.wiley.com/doi/10.1002/anie.4046829