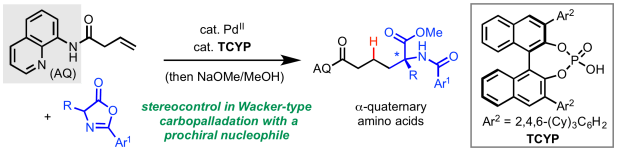
The third and final birthday preprint is now online in ChemRxiv. In this manuscript we report a previously unexplored mode of stereoinduction in Wacker type nucleopalladation—namely, desymmetrization of prochiral carbon-nucleophiles. The new method employs the dual action of palladium(II) to activate the alkene and a chiral phosphoric acid (CPA) to provide stereoinduction in the enantioselectivity-determining protodepalladation step. Congrats to the authors, Sri, Mingyu, Malkanthi, De-Wei, and Omar, as well as collaborators Jason Chen from the Automated Synthesis Facility at Scripps Research and Peng Liu from University of Pittsburg. Nice work! For a link to the manuscript, click here.