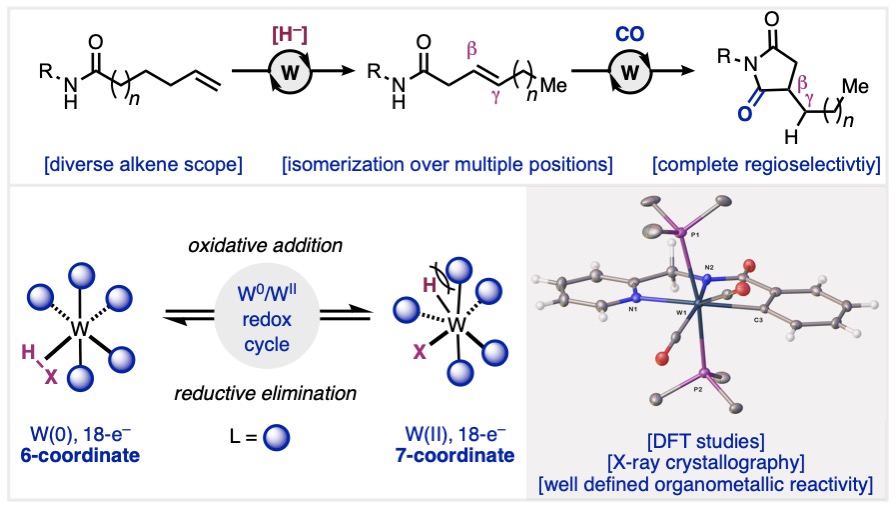
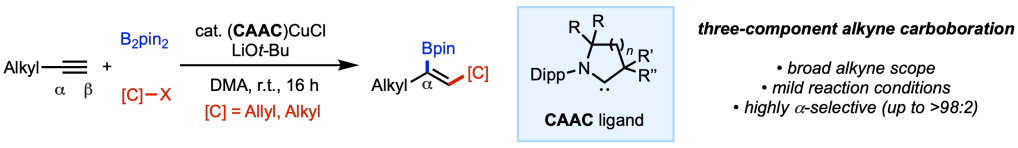
Over the years, we’ve found that translating lessons learned in alkene difunctionalization to alkyne substrates is not as straightforwards as it seems, owing to unique reactivity profiles of alkyl- versus alkenylpalladium species. In our most recent collaboration with the Shi group from Zhejiang University and the Lan group from Zhengzhou University, we show that use of the 2-(pyridin-2-yl)isopropyl (PIP) amide directing group enables regio- and stereoselective oxyhalogenation of alkynes. The alkenyl halide products can be readily diversified via cross-coupling to access an array of tetrasubstituted alkene products. Congrats to Mingyu, Juntao, and Raul (Scripps Research); Tao (Zhengzhou U), Yi and Ye-Qiang (Zhejiang U) for an excellent study.
For a link to the ChemRxiv pre-print, click here: https://chemrxiv.org/engage/chemrxiv/article-details/62a263752e62696e467d88b5