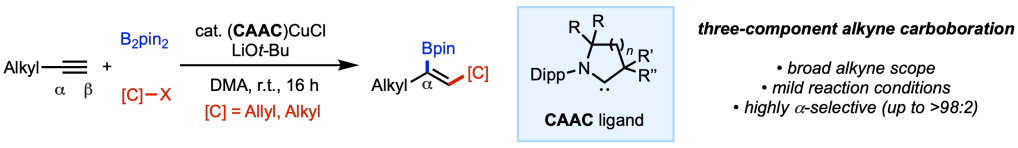
The final version of our collaborative study with the Grotjahn lab at SDSU and the Bertrand lab at UCSD has been accepted for publication in ACS Catalysis and appears online this week. We find that under (CAAC)Cu catalysis (CAAC = cyclic (alkyl)(amino)carbene), three-component carboboration of terminal alkynes takes place to furnish branched alkenyl boron compounds with high levels of regioselectivity. Building on our prior findings, we show the CAAC ligands can override substrate control factors and dictate regioselectivity of borylcupration with various carbon electrophiles, including alkyl iodides and allyl alcohol derivatives. Congrats to the entire project team: Yang, Nana, Skyler, Mingyu, and Aaron from our lab; Sima and Doug from SDSU, and Rudy and Guy from UCSD.
For a link to the paper in ACS Catalysis, see: https://pubs.acs.org/doi/full/10.1021/acscatal.2c00614
As a reminder, a pre-print of the paper was deposited in ChemRxiv in February: https://chemrxiv.org/engage/chemrxiv/article-details/61f8b9da360c842812b1d6c2
