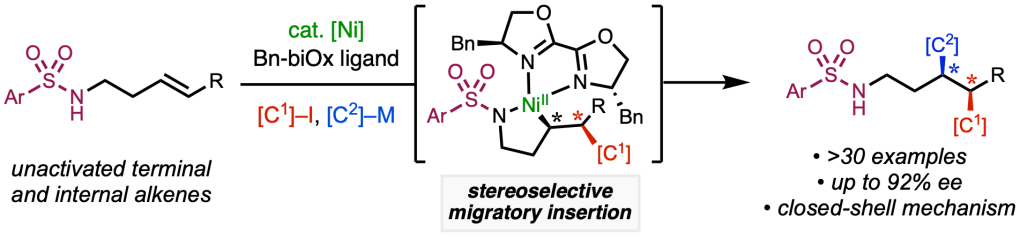
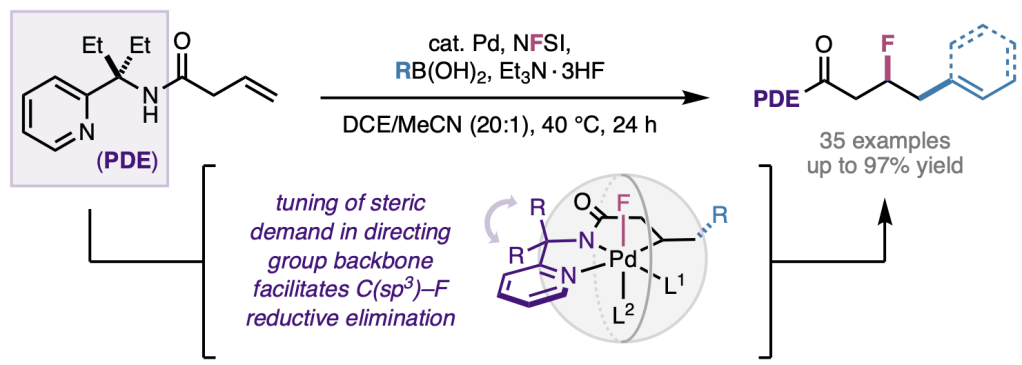
Our recent study describing the palladium(II)-catalyzed coupling of nitroalkanes and unactivated alkenes has been accepted and appears online today in ACS Catalysis. We describe how the combination of PdI2 as catalyst and HFIP as solvent is uniquely effective in enabling this historically challenging coupling process. The method allows for simple readily accessible starting materials to be elaborated into densely functionalized nitroalkane products, which can then be further derivatized as desired. Teaming up with our computational collaborators from the Liu group at the University of Pittsburgh, we describe how the carbopalladation transition state is stabilized by a Na···I interaction and an H···I hydrogen bond with HFIP. Congrats to Amit, Por, Hui-Qi, and John from our group; and Turki and Binh from the Liu group on this study.
To read the peer-reviewed paper in ACS Catalysis, click here: https://pubs.acs.org/doi/10.1021/acscatal.2c04557
As a reminder, a pre-print describing this transformation first appeared in ChemRxiv back in February: https://chemrxiv.org/engage/chemrxiv/article-details/61f8af581fd27415ef2206d7