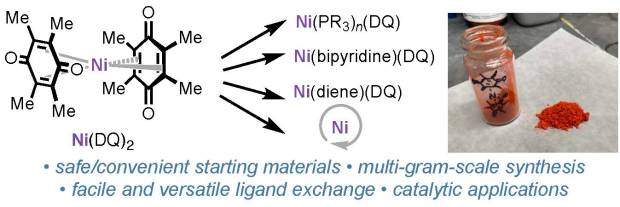
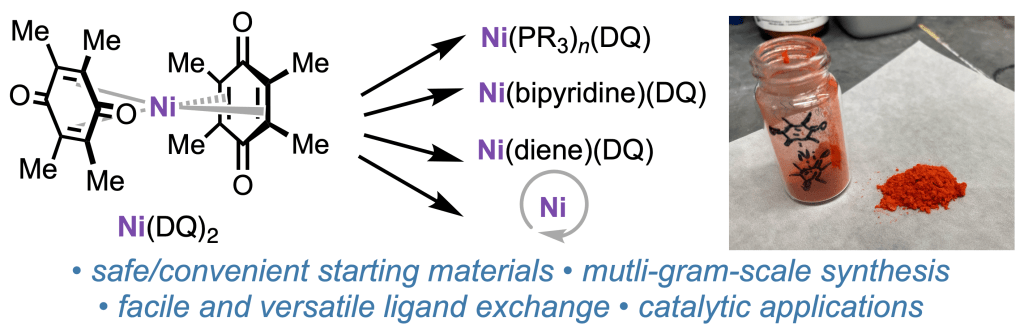
We’re excited to announce that the manuscript previously shared as a ChemRxiv pre-print—“Ni(DQ)₂: A Useful Gateway to Zero-Valent Nickel Complexes”—has now been accepted for publication in Organometallics.
In this work, we teamed up with collaborators at Bristol Myers Squibb, UCSD, and Cornell to develop a practical and scalable approach to synthesize Ni(DQ)₂ (DQ = duroquinone) from readily available nickel precursors, guided by DFT calculations. This method provides an accessible entry to zero-valent nickel complexes, which are valuable intermediates in organometallic synthesis and catalysis.
Huge congratulations to all authors and collaborators! This publication builds on the exciting progress our group has been making in air-stable nickel pre-catalysts.
Stay tuned for more updates as additional manuscripts from our group move through peer review!
📄 Read the paper in Organometallics: https://pubs.acs.org/doi/abs/10.1021/acs.organomet.5c00450
🧪 Original preprint on ChemRxiv (November 2025): https://chemrxiv.org/doi/10.26434/chemrxiv-2025-z69d2