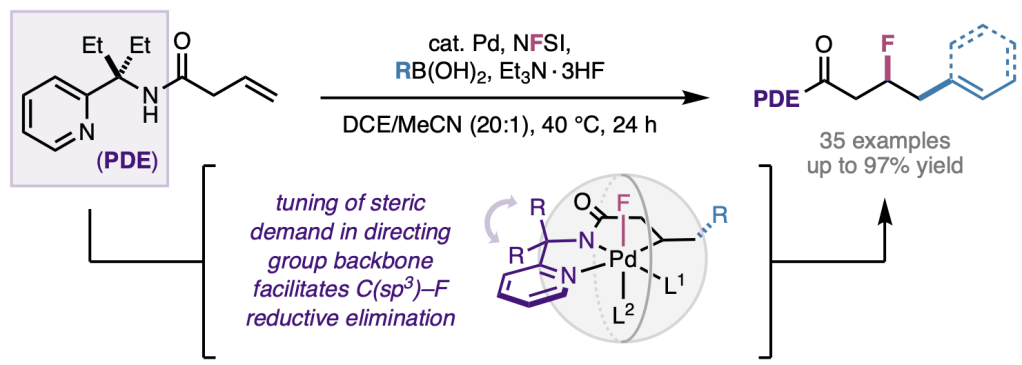
In the first of two papers appearing in final peer-reviewed form today, we report a new method for 1,2-carbofluorination of unactivated alkenes under Pd(II)/Pd(IV) catalysis in Angewandte Chemie International Edition. This new method is enabled by a unique sterically bulky gem-diethyl pyridyl amine directing group that coordinates to the palladium(II) catalyst in a bidentate fashion, controlling the regioselectivity of migratory insertion and promoting C(sp3)–F reductive elimination. The method can tolerate different substitution patterns of the alkene as well as a broad scope of aryl- and alkenylboron coupling partners. Congrats to entire team: Zhonglin, Luke, Juntao, Zi-Qi, and Nana from Scripps; and Geraint from BMS Discovery Chemistry.
For a link to the paper in Angewandte Chemie International Edition, click here: https://onlinelibrary.wiley.com/doi/10.1002/anie.202214153
As a reminder, this work was first described in a ChemRxiv pre-print back in April: https://chemrxiv.org/engage/chemrxiv/article-details/62608592d048ed03304b657d