Over the Easter weekend, Keary joined the Synthesis Workshop Podcast, hosted by Aidan Looby of the University of Houston, for a wide-ranging discussion of chemistry, career development, and educational experiences. Click below to listen to the conversation.
Category: Home
Yeongyu completes his postdoc
Camille wins 2024 WCC Merck Research Award
Congrats to our superstar G4 student, Camille Rubel, who has been selected as a recipient of a 2024 Women Chemists Committee (WCC) Merck Research Award, administered by the American Chemical Society (ACS). This nationally competitive award recognized Camille’s stellar research contributions in homogeneous nickel catalysis and her leadership in community outreach through the Barrio Logan College Institute and other initiatives. As part of the award, Camille will present her research in a special symposium at the fall ACS National Meeting in Denver, CO. Congrats Camille!
For more about the award, click here: https://acswcc.org/awards/merck-research-award/
New Collaborative Review on Industrially Relevant non-PGM Catalytic Reactions – Pre-Print Online
Together with colleagues at AbbVie and SinoCompound, we are pleased to present a pre-print describing the growing role of non-platinum-group metals (non-PGMs) in industrial manufacturing. In putting together this review, it’s been awesome to be able to bring together expertise from academia, process chemistry, and catalyst manufacturing.
For a link the pre-print in ChemRxiv, click here: https://chemrxiv.org/engage/chemrxiv/article-details/65fd5179e9ebbb4db940d941
Nickel-Catalyzed Stereodivergent Olefin Isomerization – In Press in Angewandte Chemie
The final peer-reviewed version of our nickel-catalyzed stereodivergent olefin isomerization paper appears online today in Angewandte Chemie. High Z-selectivity is facilitated by the combination of a bisphosphine ligand and an electron-deficient aryl iodide additive, which serves as a redox buffer to ensure the nickel catalyst is held in the +2 oxidation state. On the other hand, high E-selectivity is enabled by the use of a bulky trialkylphosphonium salt, which functions as the ligand and initial hydride source. Congrats to all of the authors, Camille, Anne, Calvin, Shenghua, and Zi-Qi, and thanks to our fantastic collaborator Julien of Syngenta!


For a link to the paper in Angew. Chem. Int. Ed., click here: https://onlinelibrary.wiley.com/doi/10.1002/anie.202320081
As a reminder, a ChemRxiv pre-print on this work was uploaded in December 2023: https://chemrxiv.org/engage/chemrxiv/article-details/658b9e9766c13817291f94af
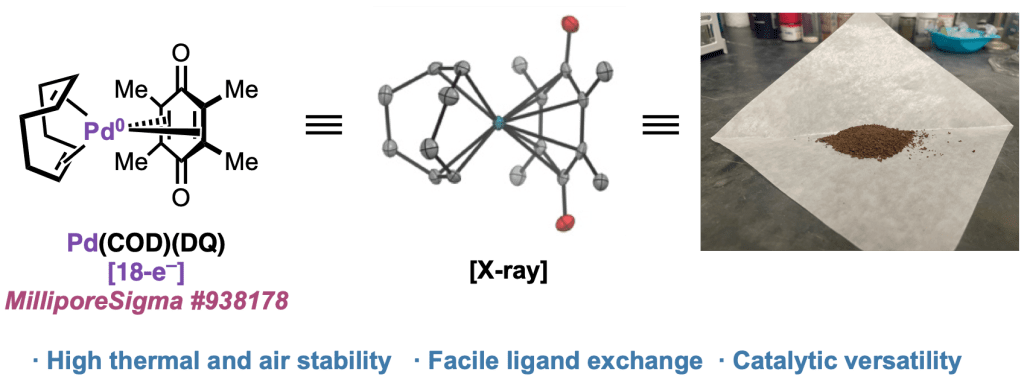
New Bench-Stable Palladium(0) Precatalyst – Pre-print online
In a ChemRxiv pre-print appearing today, we present the development of Pd(COD)(DQ), a highly stable Pd(0) source that can be used in organometallic synthesis and catalysis. We describe an operationally simple synthesis that can be performed on decagram scale; demonstrate ligand exchange with numerous phosphine, nitrogen, and NHC ligands; and showcase a variety of catalytic carbon–carbon and carbon–heteroatom coupling reactions. Pd(COD)(DQ) offers numerous benefits compared to the most widely used Pd(0) source, Pd2(dba)3, that we anticipate will make it a valuable addition to the synthetic toolkit. Congrats to the collaborative team from Scripps Research, UCSD, and Bristol Myers Squibb!
Check out the pre-print here: https://chemrxiv.org/engage/chemrxiv/article-details/65e8f798e9ebbb4db918a09f
New Postdoc Ester joins the lab!
Wen-Zhuo and Chen-Xi complete their internships
After a fantastic half-year, Wen-Zhuo and Chen-Xi wrapped up their undergrad internships with stellar presentations this week. Next, they will head back to Tianjin, China, to complete their senior years at Nankai University. Thanks to both of you for incredible contributions to research in the Engle lab. It will be wonderful to watch both your careers in chemistry unfold! Good luck in grad school and beyond.




Review on Bench-Stable Nickel(0) Precatalysts Online
Appearing this week in Accounts of Chemical Research, we chronicle our lab’s journey developing air- stable nickel(0) precatalysts through organometallic investigations of electron-deficient diene (EDD) ligands. This long-standing collaboration between our lab and chemists at Bristol Myers Squibb has yielded several commercially available precatalysts that are widely used in industry and academia. Congrats to the authors, Camille, Wen-Ji, and Steve (from BMS), and thanks to all of the coworkers over the years whose contributions are featured in this article.
For a link to the article, click here: https://pubs.acs.org/doi/10.1021/acs.accounts.3c00638
Decarboxylative Arylation to Prepare Quarternary Centers – Collaborative Study Now in Press at Angewandte Chemie
Today, our collaborative study with the Baran lab and colleagues from Bristol Myers Squibb, Biogen, Leo Pharma, and Enamine describing a new nickel/electrocatalytic decarboxylative arylation method to access quaternary centers appeared online in Angewandte Chemie, International Edition. The method uses a unique combination of pyridine and BINAP as ligands to enable an otherwise challenging coupling event. Camille from our lab contributed to elucidating the complex mechanism of this process. We had a blast collaborating with the team on this creative and useful method!
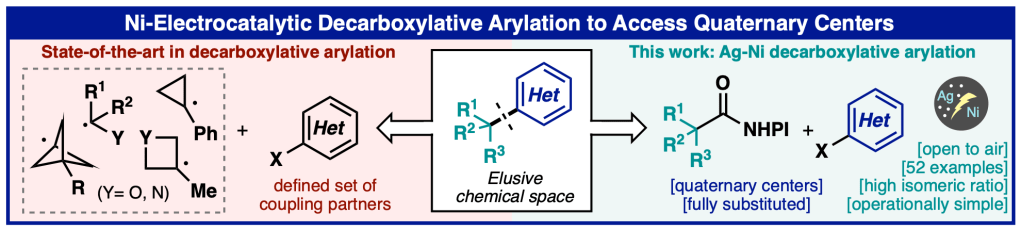
For a link to the paper in Angewandte Chemie, International Edition, click here: https://onlinelibrary.wiley.com/doi/10.1002/anie.202314617
As a reminder, a ChemRxiv pre-print on this work was uploaded in September 2023: https://chemrxiv.org/engage/chemrxiv/article-details/64f248dddd1a73847ffb6e0d