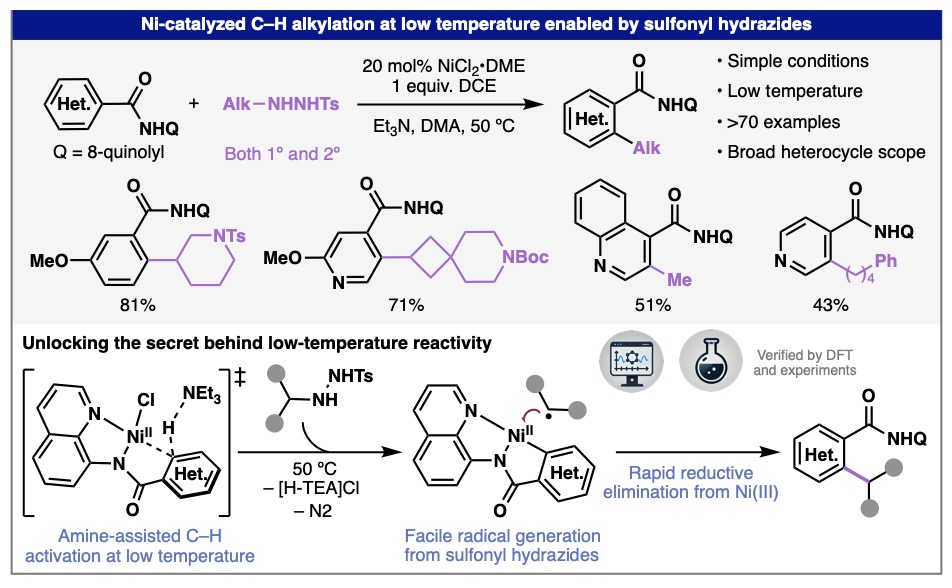
In an exciting new collaboration with the Baran lab, we find that the combination of alkylsulfonyl hydrazides and nickel catalysis unlocks unprecedented breadth in C(sp2)–C(sp3) cross-coupling via (hetero)aryl C–H activation. The method enables rapid synthesis of valuable attached ring motifs from minimally functionalized starting materials. Through a combination of experimental and computational techniques, we pinned down a unique C–H activation mechanism, termed amine-assisted asynchronous metalation/deprotonation, where the amine base used for radical generation plays a second role in mediating C–H cleavage. Congrats to the team led by Shuanghu and David from the Baran lab and Yilin from our group!
For a link to the pre-print in ChemRxiv, click here: https://chemrxiv.org/engage/chemrxiv/article-details/67e0130e6dde43c908ed25da