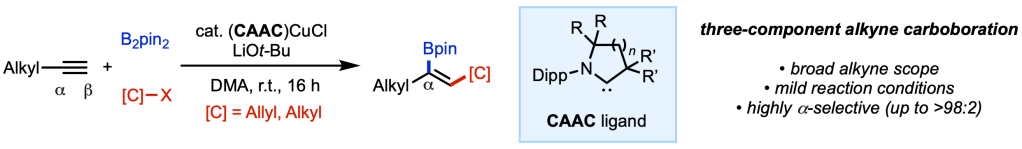
The latest results from our collaboration on (CAAC)Cu catalysis (CAAC = cyclic(alkyl)(amino)carbene) with the Grotjahn lab (SDSU) and Betrand lab (UCSD) are described in a pre-print appearing today in ChemRxiv. Building on our previous discovery that strong σ-donating CAAC ligands can effectively quench the Lewis acidity of boryl ligand on the Cu center and promote regioselective boryl transfer to the internal (α) position (Angew. Chem. Int. Ed. 2021, 60, 19871) in protoboration, in the current study we extend this concept to a number of C(sp3) electrophiles. This protocol allows for expedient and selective assembly of alkenylboron products that are versatile building blocks in synthesis. Hats off to the tri-institutional team: Yang, Nana, Sima, Mingyu, Aaron, Doug, Guy, Rudy, and Keary!
For a link to the ChemRxiv pre-print, click here: https://chemrxiv.org/engage/chemrxiv/article-details/61f8b9da360c842812b1d6c2