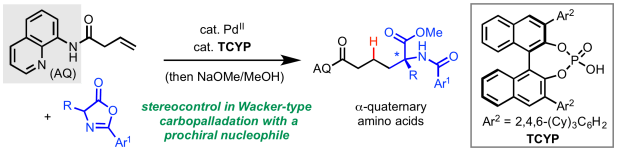
For the past two years the Engle lab has been hard at work developing effective strategies to control enantioselectivity in catalytic directed alkene hydrofunctionalization and 1,2-difunctionalization. Today, a new method for enantioselective α-alkylation of azlactones with non-conjugated alkenes via Pd(II)/chiral phosphoric acid catalysis was accepted for publication in Angew. Chem. Int. Ed. (click here). This unique transformation enables preparation of highly functionalized quaternary α-amino acids. In collaboration with Prof. Peng Liu, we performed a series of detailed experimental and computational studies to elucidate a viable model to explain stereoinduction in which the CPA plays the dual role of coordinating to the incoming carbon nucleophile during reversible carbopalladation and then serving as a proton transfer mediator in the turnover-limiting protodepalladation step. Congrats to all of the authors from the Engle lab—Sri, Mingyu, Malkanthi, De-Wei, and Omar! Thanks too to Prof. Liu and Dr. Jason Chen from the Automated Synthesis facility at Scripps Research. In case anyone missed it, a pre-print of this work was published on ChemRxiv back in early November (click here).