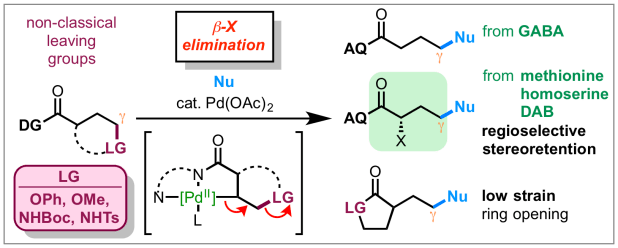
In a paper appearing today online in Nat. Chem., the Engle lab reports a method of cleaving typically unreactive carbon–carbon and carbon–heteroatom bonds via β-X elimination from alkylpalladium(II) intermediates. This reaction enables functional group metathesis (or exchange) between C(sp3)–C, C(sp3)–N, C(sp3)–O, and C(sp3)–S, and C(sp3)–F bonds, which has enabled the development of synthetically useful new transformations, such as the upgrading of inexpensive natural amino acids to valuable non-natural counterparts. Congrats to the project team of Van, John, and Kin on this awesome piece of work. Click here for a link.
 (not pictured: Kin)
(not pictured: Kin)
