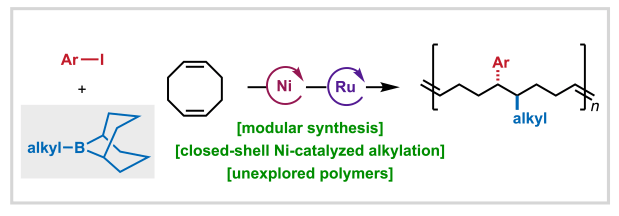
We’re pleased to share that Anne, Aimee, Shijia, and Ethan’s study developing a nickel-catalyzed 1,2-alkylarylation method for accessing sequence encoded monomers has been published in ACS Central Science!
In this report, we describe how B-alkyl-borabicyclo[3.3.1]nonane (Alkyl-9-BBN) reagents are uniquely effective for alkyl group transfer in closed-shell Ni(0)/Ni(II) cycles, enabling selective functionalization of cyclic dienes without competitive ring closure. This work exemplifies how innovative reaction design combined with mechanistic understanding can expand the frontiers of selective chemical synthesis and catalysis.
Congratulations to all co-authors and collaborators from Scripps Research, University of Pittsburgh, Georgia Tech, and Bristol Myers Squibb for this accomplishment! This publication continues to underscore our lab’s dedication to developing catalytic methods with broad utility in organic synthesis and materials chemistry.
📄 Read the paper in ACS Central Science: https://pubs.acs.org/doi/10.1021/acscentsci.5c02173
🧪 Original preprint on ChemRxiv (October 2025): https://chemrxiv.org/engage/chemrxiv/article-details/68e901addfd0d042d1dace4c